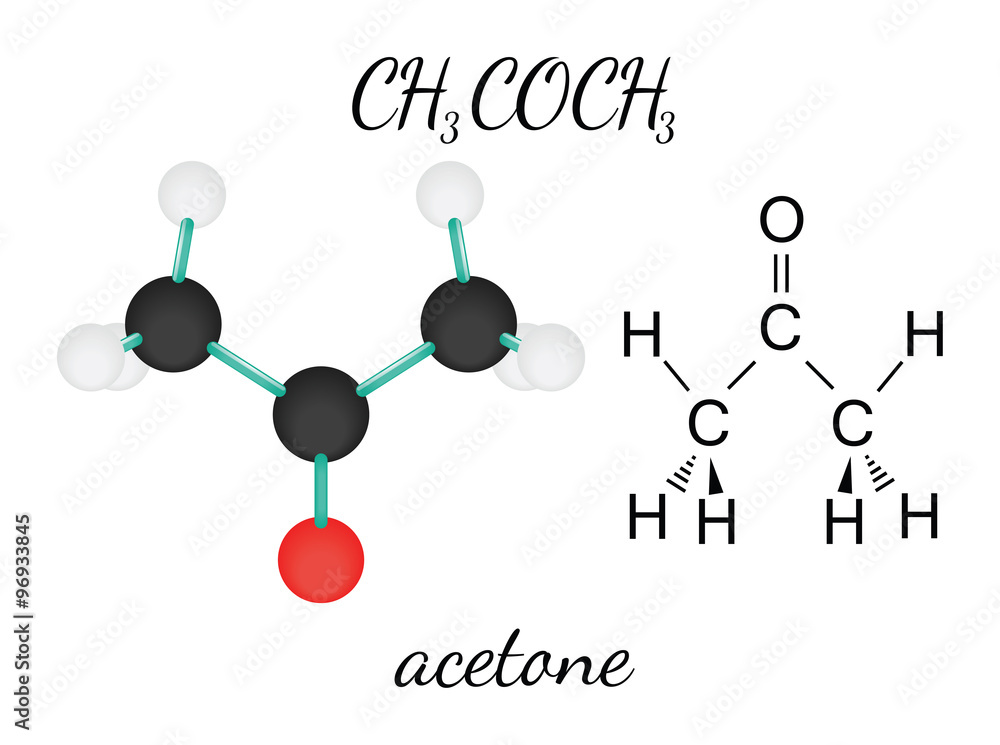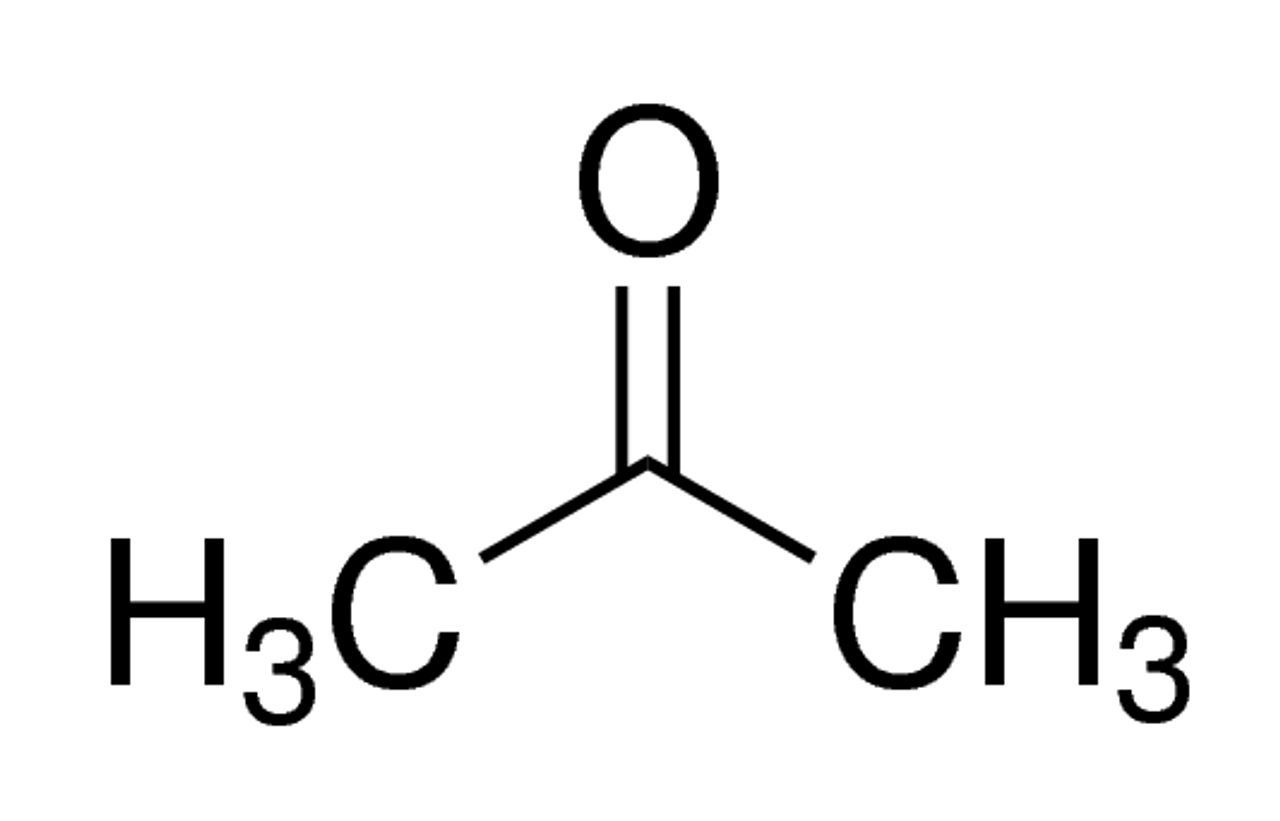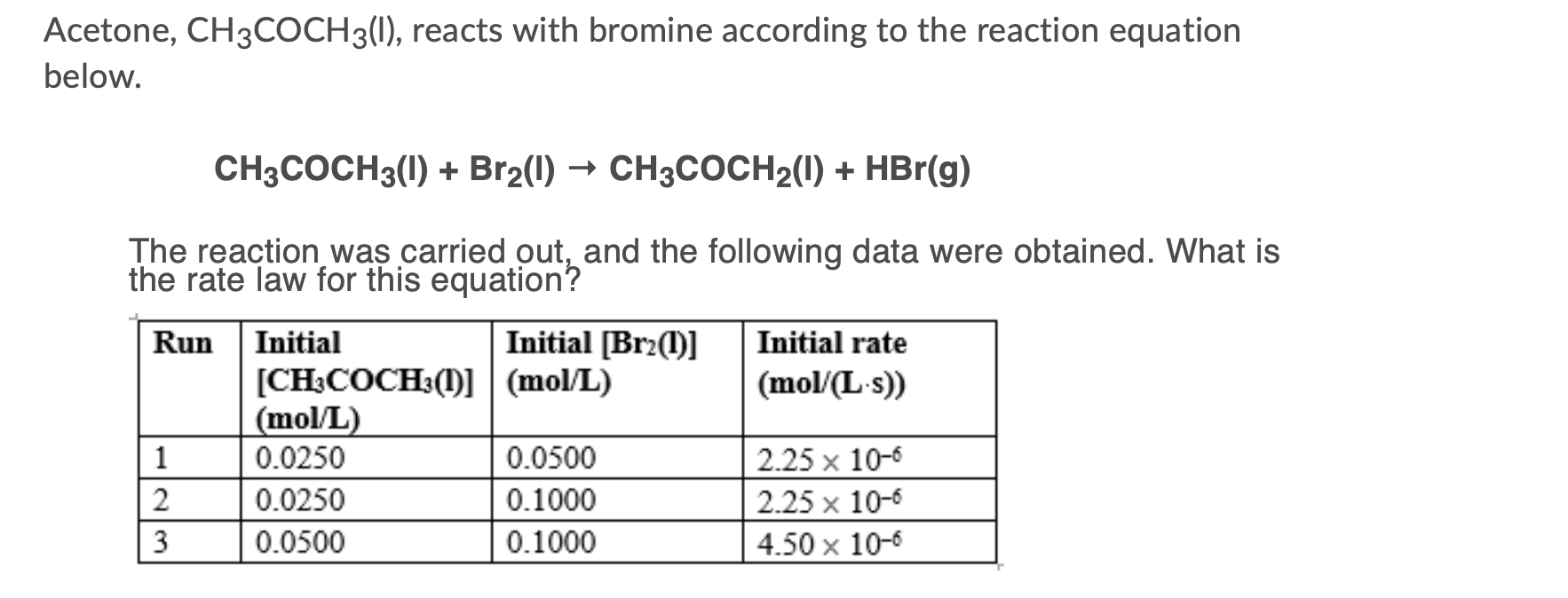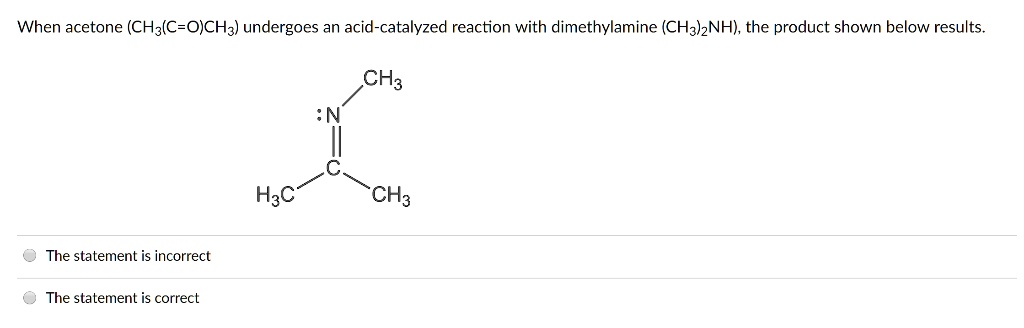
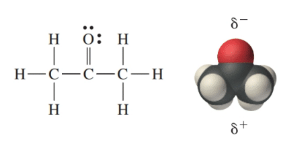
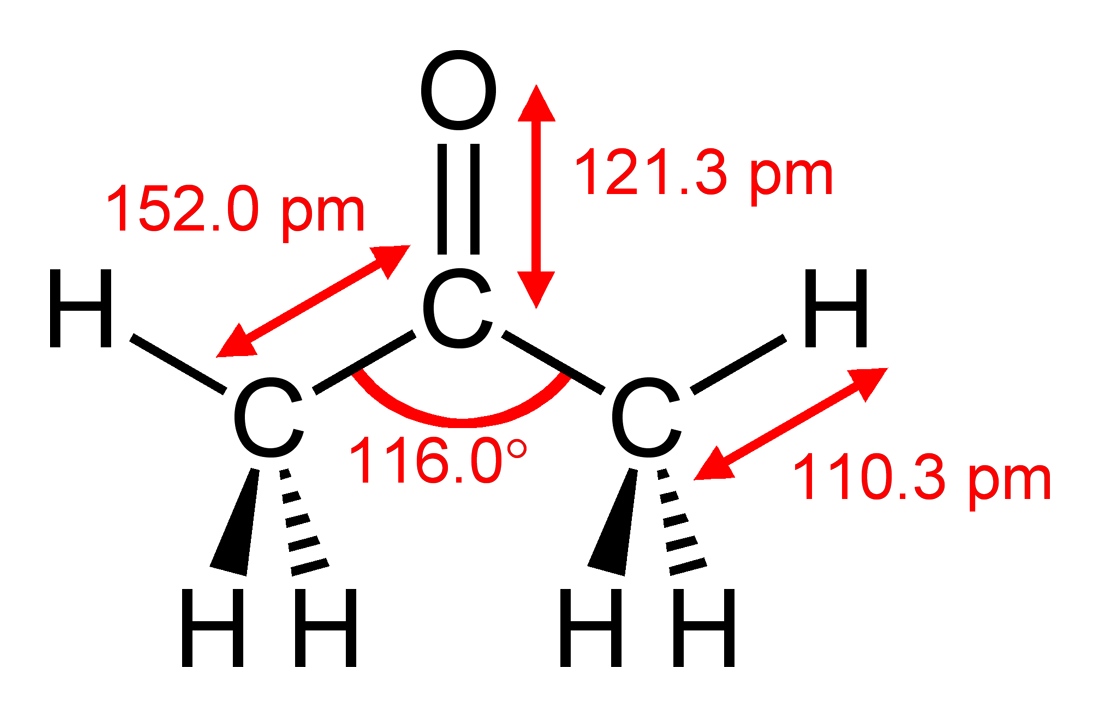
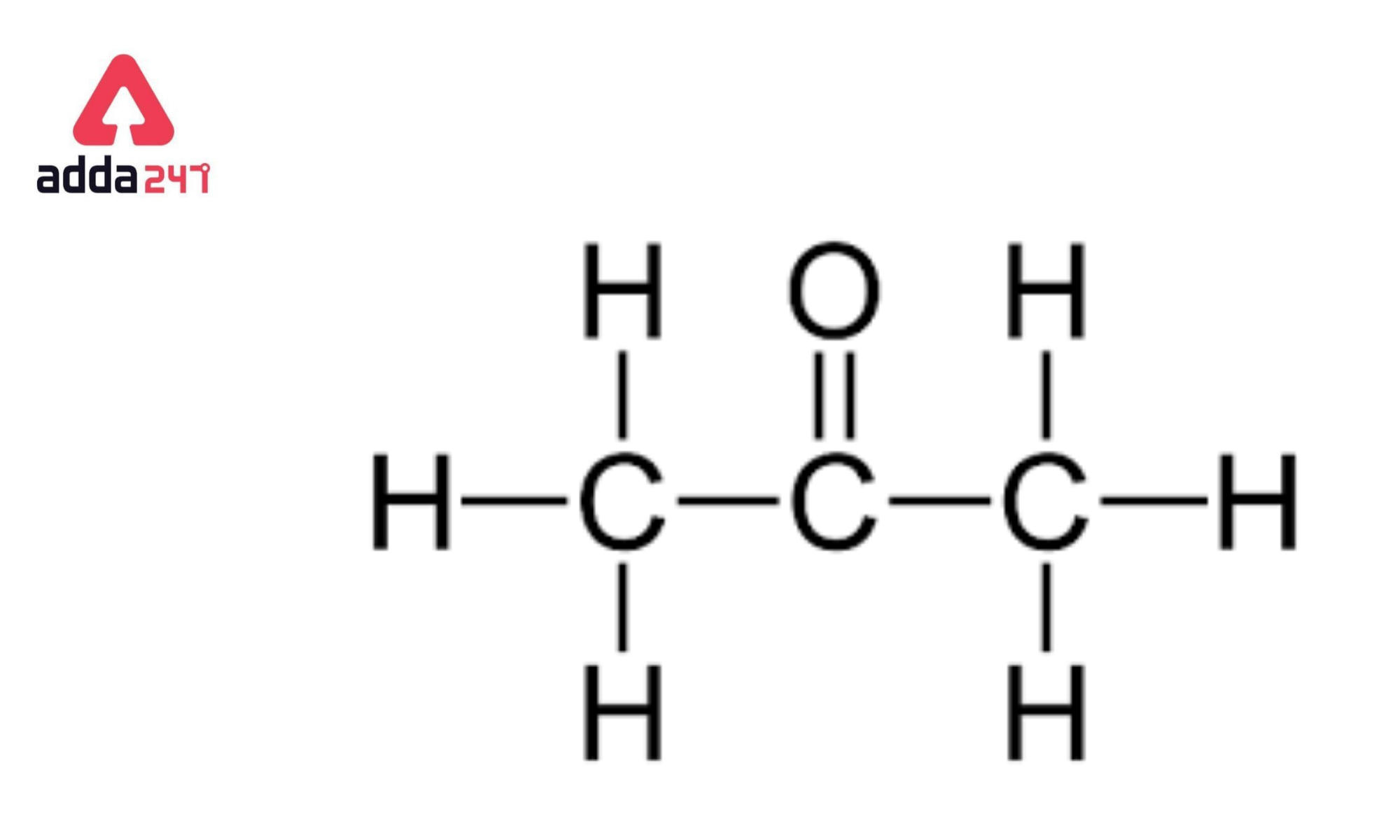
SOLVED: When acetone (CH3COCH3) undergoes an acid-catalyzed reaction with dimethylamine (CH3)2NH, the product shown below results: CH3 | C | CH3 The statement is incorrect.

Figure 1 from Dissociation of acetone radical cation (CH3COCH3(+*) --> CH3CO(+) + CH3(*)): an ab initio direct classical trajectory study of the energy dependence of the branching ratio. | Semantic Scholar

Acetone ( CH3COCH3 ) 8.7g is reduced to isopropyl alcohol ( CH3CHOHCH3 ) using excess hydrogen and a palladium catalyst. a. what mass of hydrogen is consumed? b. what mass of isopropyl

Una bottiglia di propanone o acetone (CH3COCH3) come usato in un Regno Unito scuola secondaria, Londra, Regno Unito Foto stock - Alamy


![Acetone [CH3COCH3] [CAS_67-64-1] Clear Liquid 35 Lb Pail – Wintersun Chemical Acetone [CH3COCH3] [CAS_67-64-1] Clear Liquid 35 Lb Pail – Wintersun Chemical](https://www.wintersunchemical.com/cdn/shop/products/01-068-04.jpg?v=1676676047)