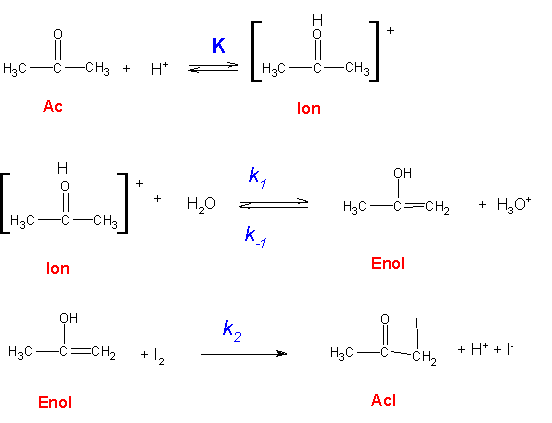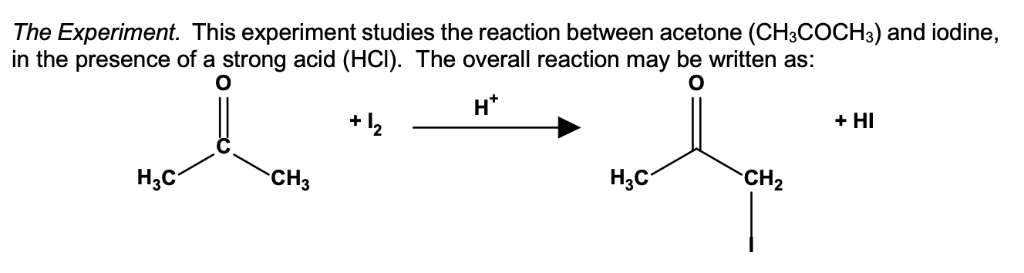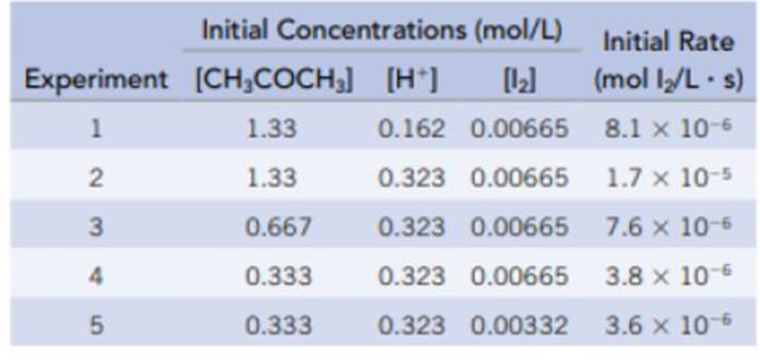
The acid-catalyzed iodination of acetone CH 3 COCH 3 (aq) + I 2 (aq) → CH 3 COCH 2 I(aq) + HI(aq) is a common laboratory experiment used in general chemistry courses

What happens when: (write equations)(a) Acetone is heated with alkaline solution of Iodine.(b) Chlorobenzene is heated with chloral in presence of conc H2SO4.(c) Chlorobenzene is heated with Sodium in presence of ether.(d)
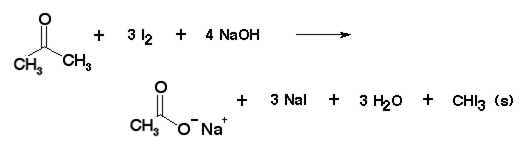
The light yellow compound produced when acetone reacts with iodine and alkali is,(A) $ C{H_3}.CO.C{H_2}I $ (B) $ C{H_3}I $ (C) $ CH{I_3} $ (D) None of these

SOLVED: The equation for the iodination of acetone in acidic solution is: CH3COCH3(aq) + I2(aq) -> CH3COCH2I(aq) + HI(aq) The rate of the reaction is found to be dependent not only on

What happens when Acetone reacts with Iodine and base - Chemistry - Aldehydes Ketones and Carboxylic Acids - 10947031 | Meritnation.com