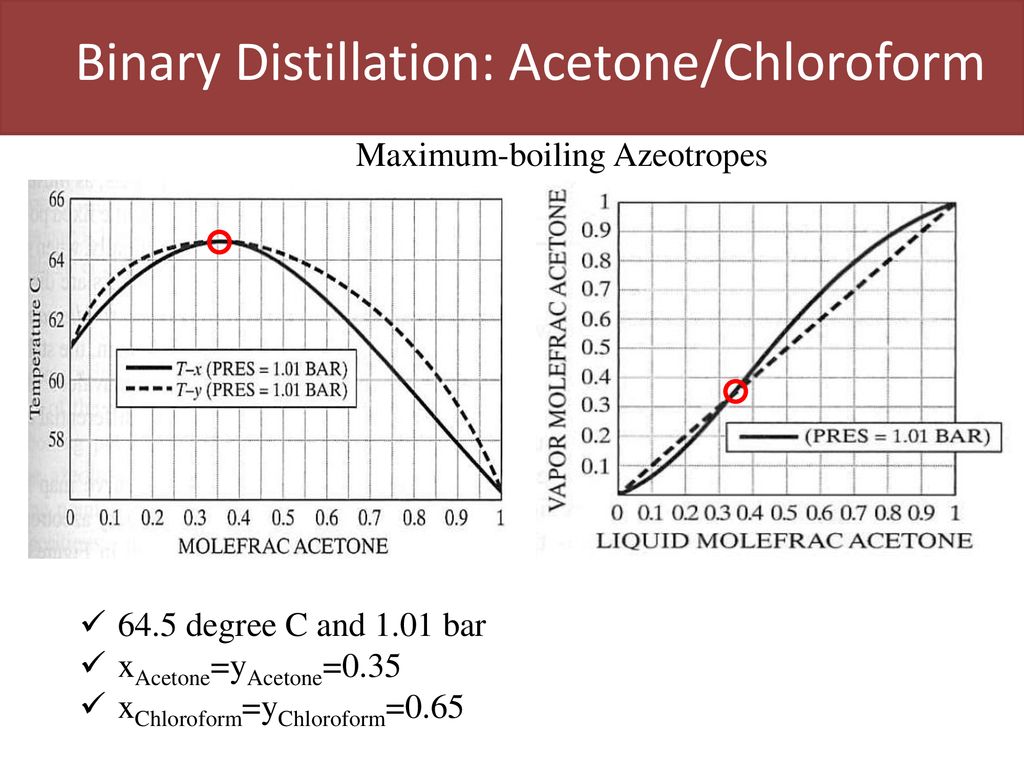
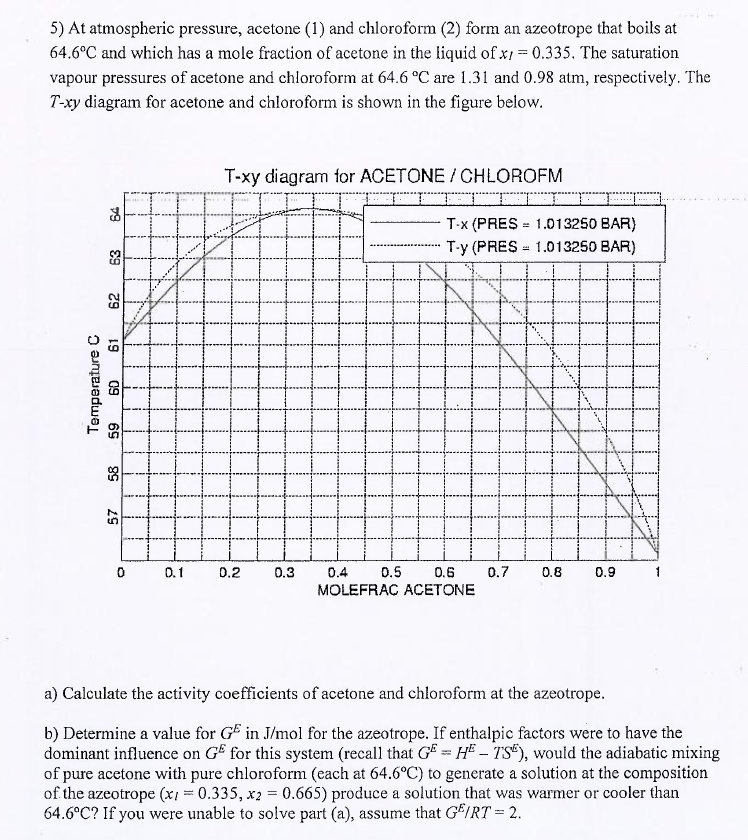
Applied Sciences | Free Full-Text | Effect of Ionic Liquids on the Isobaric Vapor-Liquid Equilibrium Behavior of Acetone-Chloroform
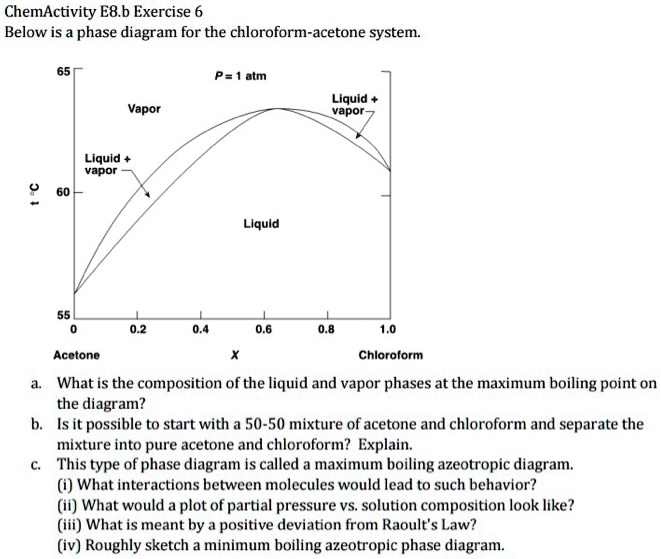
SOLVED: ChemActivity E8.b Exercise 6: Phase Diagram for the Chloroform- Acetone System Below is the phase diagram for the chloroform-acetone system: P = atm Liquid vapor Vapor Liquid vapor Liquid Acetone Chloroform What
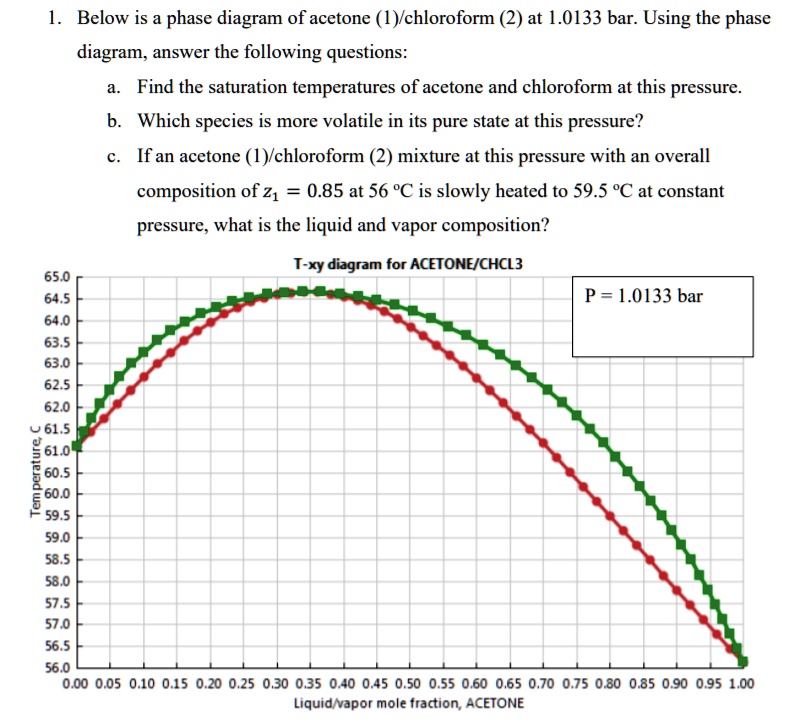
SOLVED: Below is a phase diagram of acetone (1)/chloroform (2) at 1.0133 bar. Using the phase diagram, answer the following questions: a. Find the saturation temperatures of acetone and chloroform at this
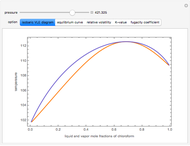
High Pressure Vapor-Liquid Equilibrium Data of a Binary Mixture of Chloroform and Acetone - Wolfram Demonstrations Project

Control of the Maximum-Boiling Acetone/Chloroform Azeotropic Distillation System | Industrial & Engineering Chemistry Research
Separation of Mixture Containing Maximum Boiling Azeotrope with Extractive Heterogeneous-Azeotropic Distillation

SciELO - Brasil - THERMODYNAMIC TOPOLOGICAL ANALYSIS OF EXTRACTIVE DISTILLATION OF MAXIMUM BOILING AZEOTROPES THERMODYNAMIC TOPOLOGICAL ANALYSIS OF EXTRACTIVE DISTILLATION OF MAXIMUM BOILING AZEOTROPES

Control of the Maximum-Boiling Acetone/Chloroform Azeotropic Distillation System | Industrial & Engineering Chemistry Research

Figure 3 from Separation of Acetone-chloroform maximum boiling azeotrope using Dimethyl sulfoxide | Semantic Scholar

Figure 2 from Separation of Acetone-chloroform maximum boiling azeotrope using Dimethyl sulfoxide | Semantic Scholar

Comparison of extractive distillation and pressure-swing distillation for acetone/chloroform separation - ScienceDirect

Table 3 from Separation of Acetone-chloroform maximum boiling azeotrope using Dimethyl sulfoxide | Semantic Scholar

QUESTION 2.37 Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of

Figure 1 from Separation of Acetone-chloroform maximum boiling azeotrope using Dimethyl sulfoxide | Semantic Scholar
Steady State Design for the Separation of Acetone-Chloroform Maximum Boiling Azeotrope Using Three Different Solvents
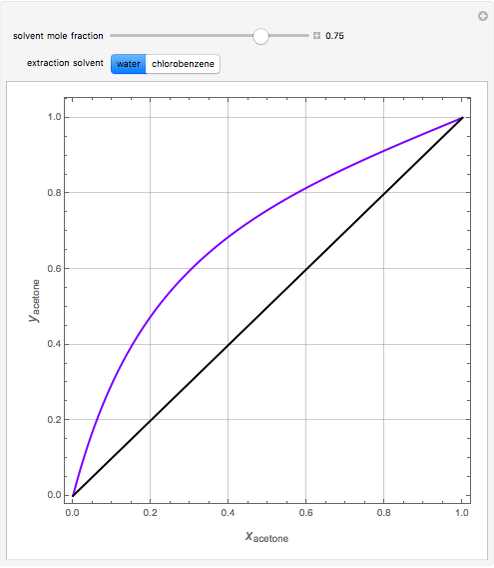
Breaking the Acetone-Methanol Azeotrope with Different Extraction Solvents - Wolfram Demonstrations Project
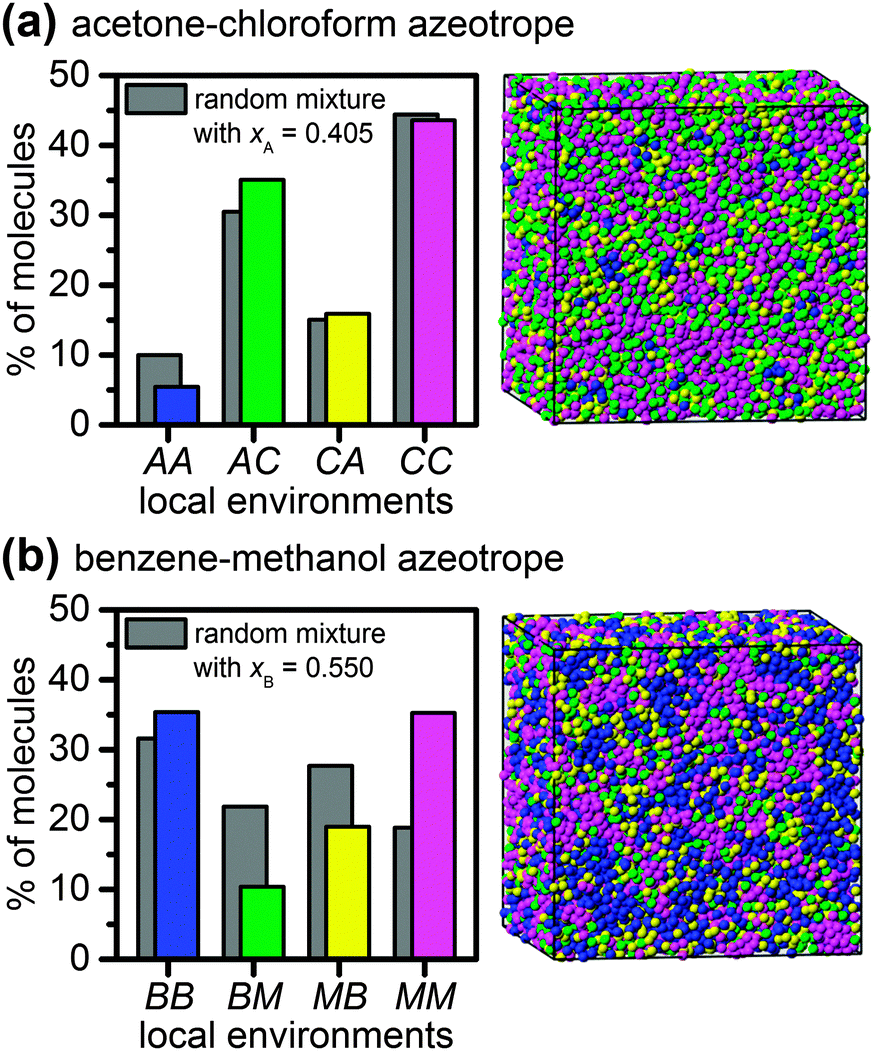
Microstructures of negative and positive azeotropes - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C6CP02450E