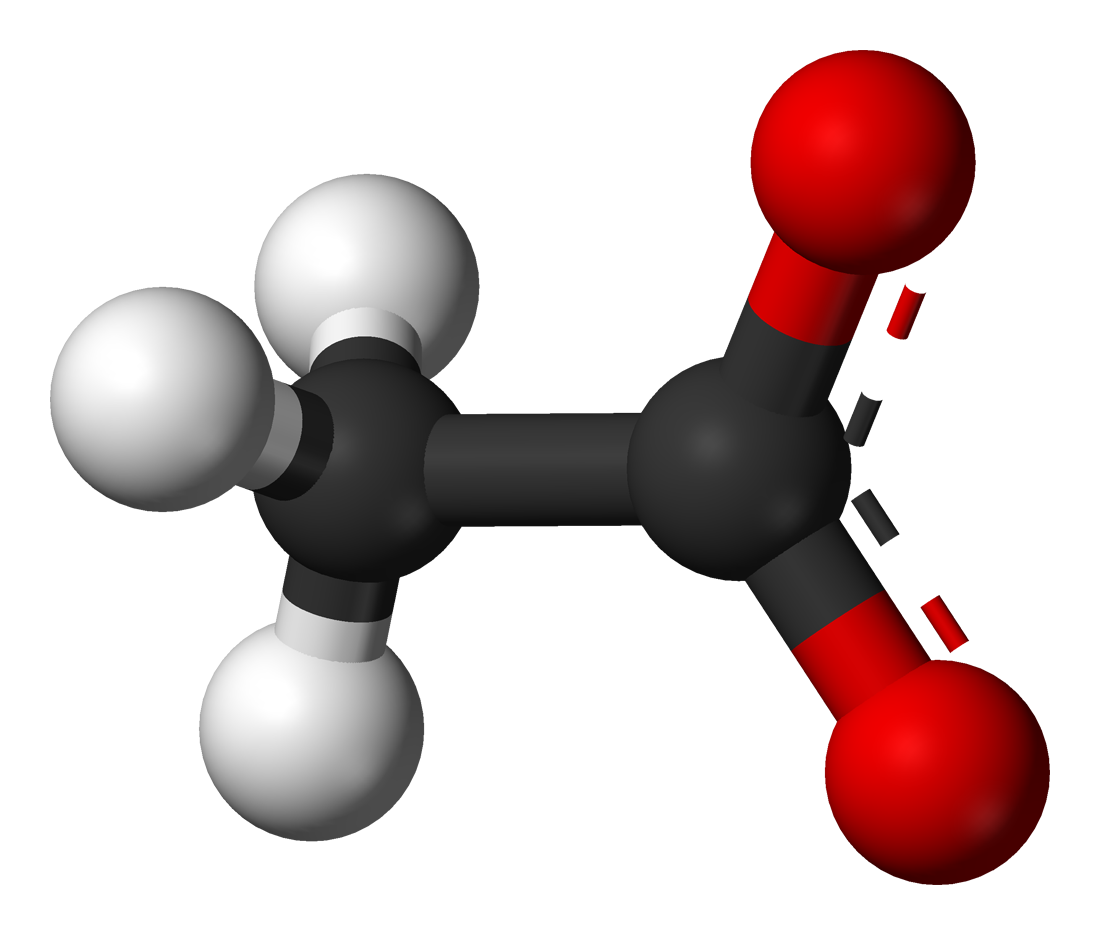
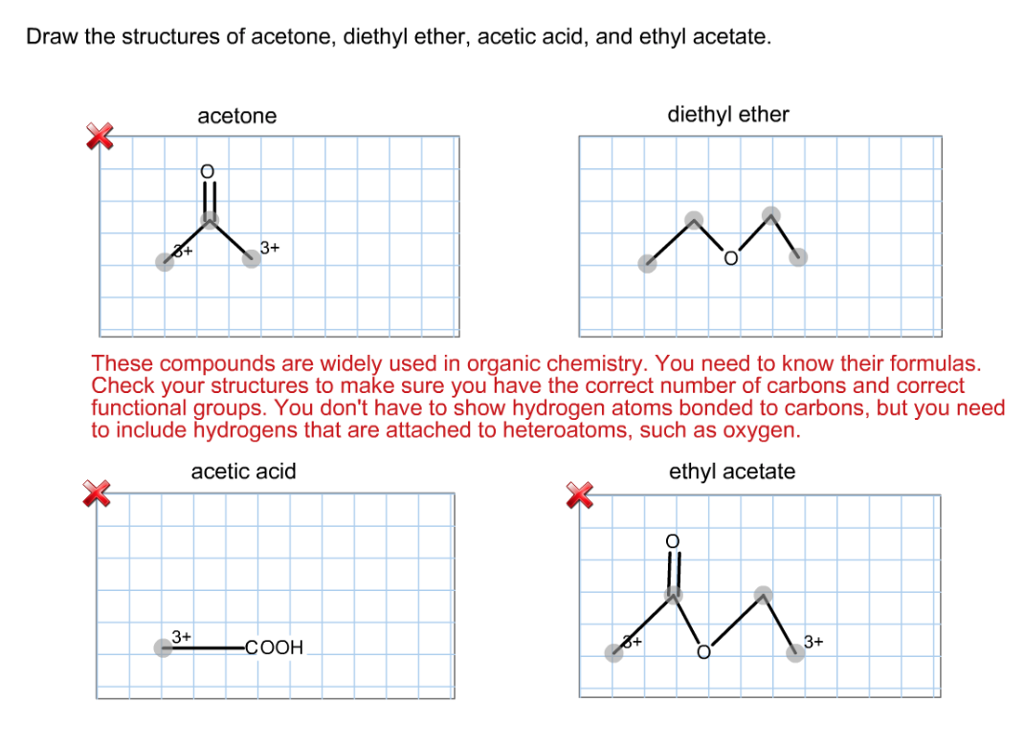
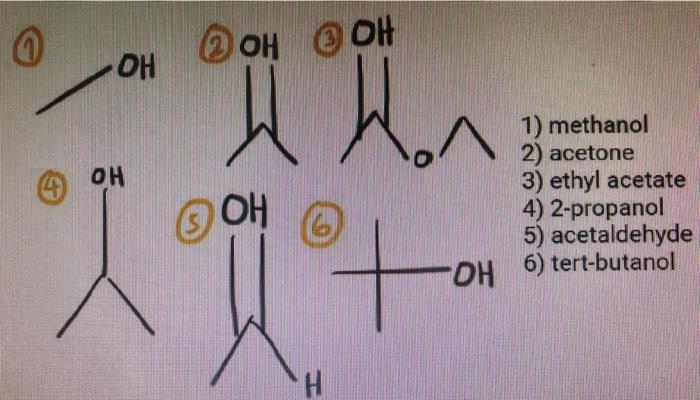
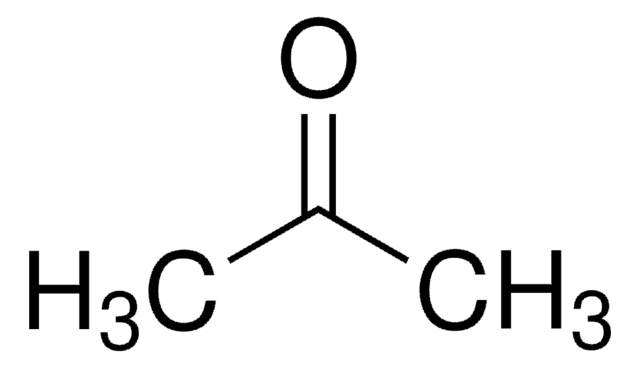
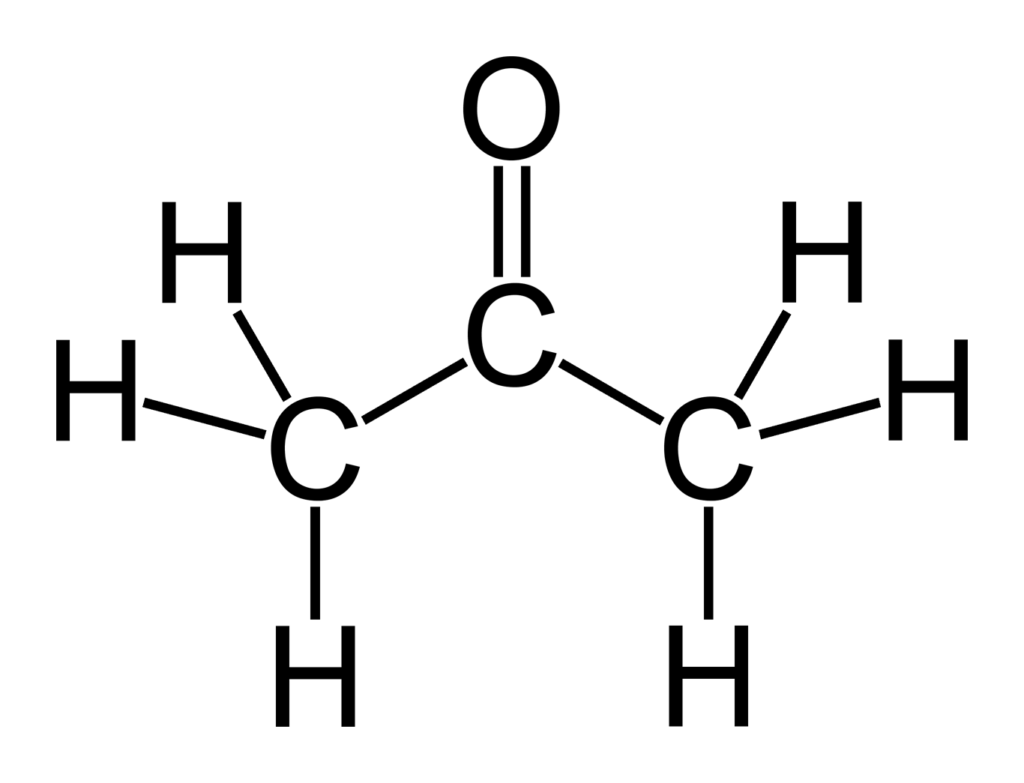
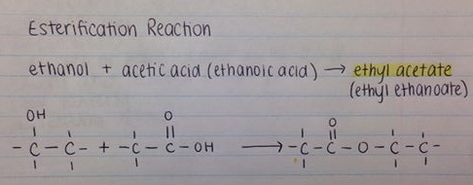What is the mechanism when acetone is treated with ethyl chloroacetate in the presence of NaNH2? - Quora
SOLVED: The boiling point for acetic acid is 118 'c, but is 56 'c for acetone. Select which answer(s) explain the difference in these values. ethyl acetate acetone tetrahydrofuran m-xylene ethanol n-hexane

SOLVED: 1) A mixture of Ethyl acetate and water is used for extracting a compound. Discuss your observations and explain why? 2) There are several practices that need to be avoided during
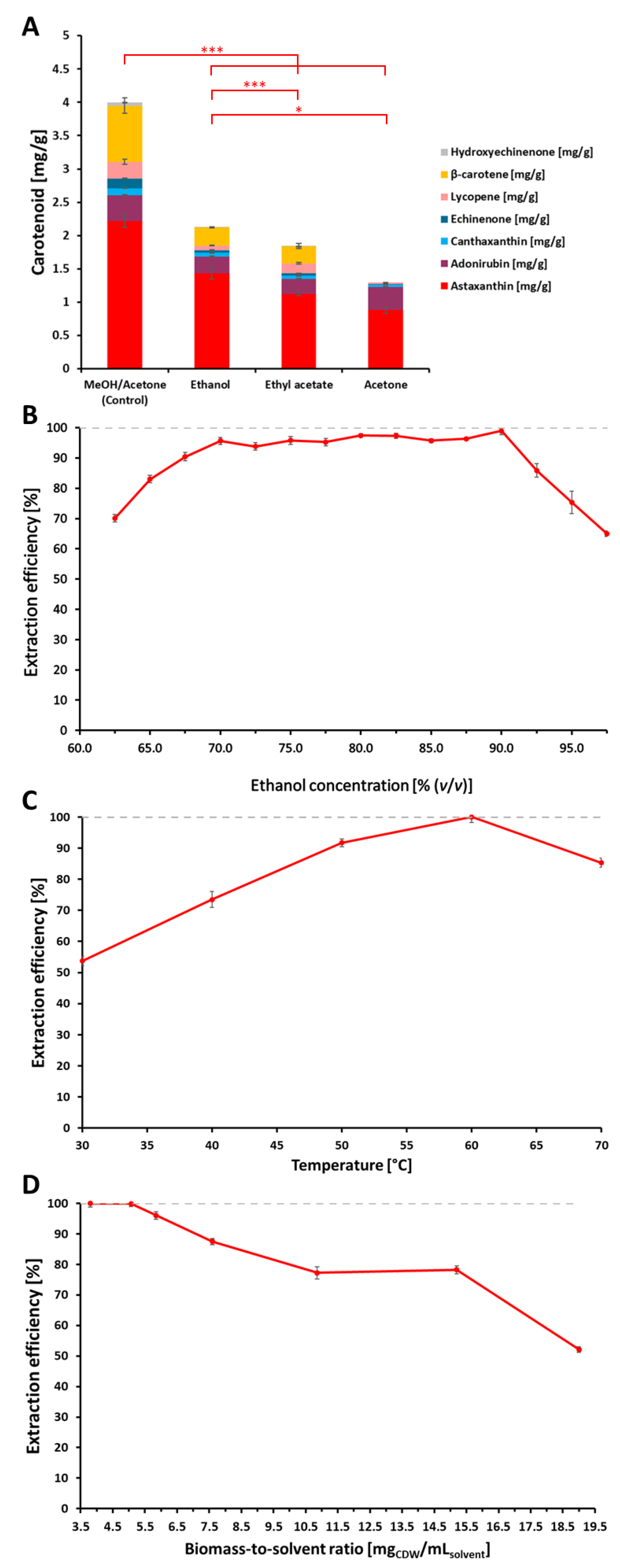
Marine Drugs | Free Full-Text | Extraction and Purification of Highly Active Astaxanthin from Corynebacterium glutamicum Fermentation Broth

Isobaric Vapor–Liquid Equilibria of Binary Mixtures of γ-Valerolactone + Acetone and Ethyl Acetate | Journal of Chemical & Engineering Data
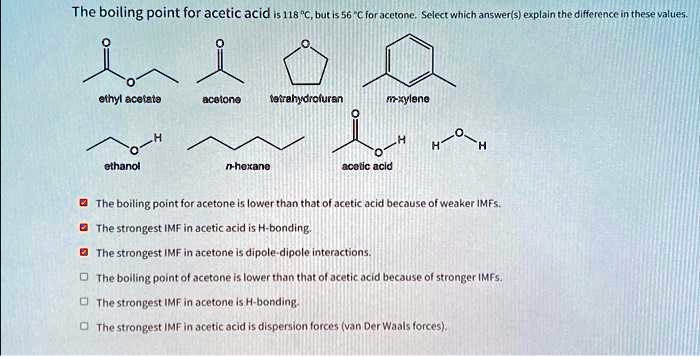
SOLVED: Texts: The boiling point for acetic acid is 11°C but is 56°C for acetone. Select which answers explain the difference in these values. 1. ethyl acetate 2. acetone 3. tetrahydrofuran 4.